I've discussed cellular intelligence before, including local intelligence, especially in the synapses of nerve cells. But now a new paper1 has added another dimension to the intelligence around the synapse, specifically the post-synaptic processes.
In Synaptic activation modifies microtubules underlying transport of postsynaptic cargo (by Christoph Maasa, Dorthe Belgardta, Han Kyu Leea, Frank F. Heislera, Corinna Lappe-Siefkea, Maria M. Magierab,
Juliette van Dijkb, Torben J. Hausrata, Carsten Jankeb, and Matthias Kneussel), we hear that synaptic activity, that is the activity of incoming action potentials, can modify a part of the cellular skeleton called microtubules in a way that changes the characteristics of the postsynaptic density, contributing to Synaptic plasticity. Synaptic plasticity is the ability of the synapse to change how it responds to an action potential, in terms of the final effect on the dendrite and its tendency to fire a new action potential.
The mechanism of this modification is in changing the level (and probably the type) of Polyglutamylation, a process in which several (or more) glutamate residues are added to an existing glutamate residue in the tubulin proteins of the microtubule.

Figure 1: Polyglutamylation and polyglycylation. (From the website of Group Carsten Janke.)
The result of this modification is that receptors for glycine, and probably GABA,2 both neurotransmitters that normally tend to suppress new action potentials, are changed in their behavior migrating to the synapse.
This process is mediated by a protein called gephyrin, a part of the cellular skeleton normally found associated with the postsynaptic density. It turns out that gephyrin is also involved in mediating the transport of vesicles3 containing glycine receptors. Apparently, changes in the polyglutamylation change the affinity of gephyrin for the transport process. Gephyrin has a strong tendency to bind to one of the monomers of the glycine receptors, which helps to stabilize these receptors in the postsynaptic density. Evidently by binding to this protein when it is in a transport vesicle, as well as to one of the molecular motors that runs along the microtubules (KIF5), it works to transport the receptors to the synapse.
This relationship was demonstrated by removing neuronal polyglutamylase, an enzyme necessary to the modification process.
What hasn't been clarified yet is the actual mechanism by which activity at the synapse modifies the polyglutamylation of microtubules, although it clearly involves neuronal polyglutamylase in some way. However, the simple fact that it does tells us a lot.
Let's start by looking at the relationship of glycine and GABA. Gephyrin can bind to receptor complexes for both neurotransmitters, so the same transport mechanism that moves glycine receptors to the synapse could well move GABA receptors. It's known2 that these neurotransmitters work together:
[... G]lycinergic and GABAergic synapses display morphological similarities. Glycine and GABA notably share the same presynaptic vesicular transporter (the vesicular inhibitory amino acid transporter VIAAT) ([refs]) while the anchoring protein gephyrin is involved in the aggregation and postsynaptic stabilization of both GlyRs and GABAARs ([ref]). Accordingly, it is now well established that glycinergic and GABAergic neurotransmission can be intermingled to provide inhibition of neuronal activity. Indeed, in the spinal cord and several distinct brainstem areas, glycine and GABA can be co-released from the same presynaptic terminal to activate postsynaptic GlyRs and GABAARs, as demonstrated by recording mixed miniature inhibitory postsynaptic currents. Interestingly, recent findings indicate that the co-activation of GlyRs and GABAARs at mixed inhibitory synapses does not simply display a summation of currents mediated by both receptor subtypes, resulting from functional cross-talk between these co-aggregated postsynaptic receptors.
What does this mean for synaptic intelligence?
To begin with, at least some neurons are capable of using both glycine and GABA as neurotransmitters. While we don't know yet whether, or how, they modify the ratios of these neurotransmitters, it seems likely that the presynaptic cell as a whole can change its ratio. It may even be that the precise ratio of these two neurotransmitters can be "fine tuned" at each synapse, although there's certainly no need for it. (But note that just because we know of one perfectly good mechanism to accomplish something doesn't mean that evolution hasn't co-opted another. It all depends on which mechanism becomes available first, which depends on the random chance of mutation.)
The reason there's no need to "fine tune" glycine/GABA ratios upstream from the synapse is that the ratio of receptors can be "fine-tuned" downstream. We have a demonstration that activity at glycine receptors can modify how gephyrin transports receptors, certainly glycine and probably GABA. But there are certainly other mechanisms that can affect the local ratio of these two receptors. And we don't really know yet whether the effect on GABA receptor transport is the same as that on glycine receptors.
Whatever the mechanism of modifying the microtubules, it almost certainly involves the complex network of interacting enzymes at the postsynaptic density, which, as we've seen, is potentially quite intelligent. The modification of the microtubules makes them part of the overall network, adding a new dimension to the intelligence with which a single synapse can modify its response to incoming information based on what's already arrived.
Another question that remains to be resolved involves the dendritic arbor, and the effect of modifying the microtubules of one branch.
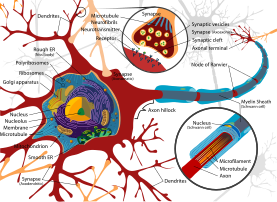
Figure 2: Complete Neuron diagram. The soma and dendrites are on the left, in red. Note how some of the synapses are on deep branches, with many synapses farther from the soma on the branch. (From Wiki)
It seems likely that modifying the transport of receptors on a "deep" branch will have some effect on all the synapses farther from the soma (the cell body). As with the effect of changes to voltage-driven channels in the dendrite, changes to the microtubules will (potentially) allow information received at synapses close to the soma to have a nonlinear effect on a large group of synapses.
This discovery adds a new dimension to the potential for intelligence in each specific synapse, as well as the overall neuron.
Maas, C., Belgardt, D., Lee, H., Heisler, F., Lappe-Siefke, C., Magiera, M., van Dijk, J., Hausrat, T., Janke, C., & Kneussel, M. (2009). Synaptic activation modifies microtubules underlying transport of postsynaptic cargo Proceedings of the National Academy of Sciences DOI: 10.1073/pnas.0812391106
1. Synaptic activation modifies microtubules underlying transport of postsynaptic cargo
2. Extrasynaptic and postsynaptic receptors in glycinergic and GABAergic neurotransmission: a division of labor?
3. Neuronal cotransport of glycine receptor and the scaffold protein gephyrin

No comments:
Post a Comment